What a lot-level COA should actually clarify.
How we think about identity, purity, batch traceability, and the small documentation habits that make a product easier to trust.
 Zhuo Yue Labs
Zhuo Yue Labs
The ZYL Journal
A tighter read on peptide verification, cold-chain operations, formulation discipline, and the systems behind a catalog built for repeatable research workflows.
Latest Dispatches
Short, practical entries from the operating edge of a research supply chain.
Reading Library
Each piece is designed to help researchers evaluate the systems around the vial, not just the label on it.

The checks that separate useful third-party analysis from decorative paperwork.
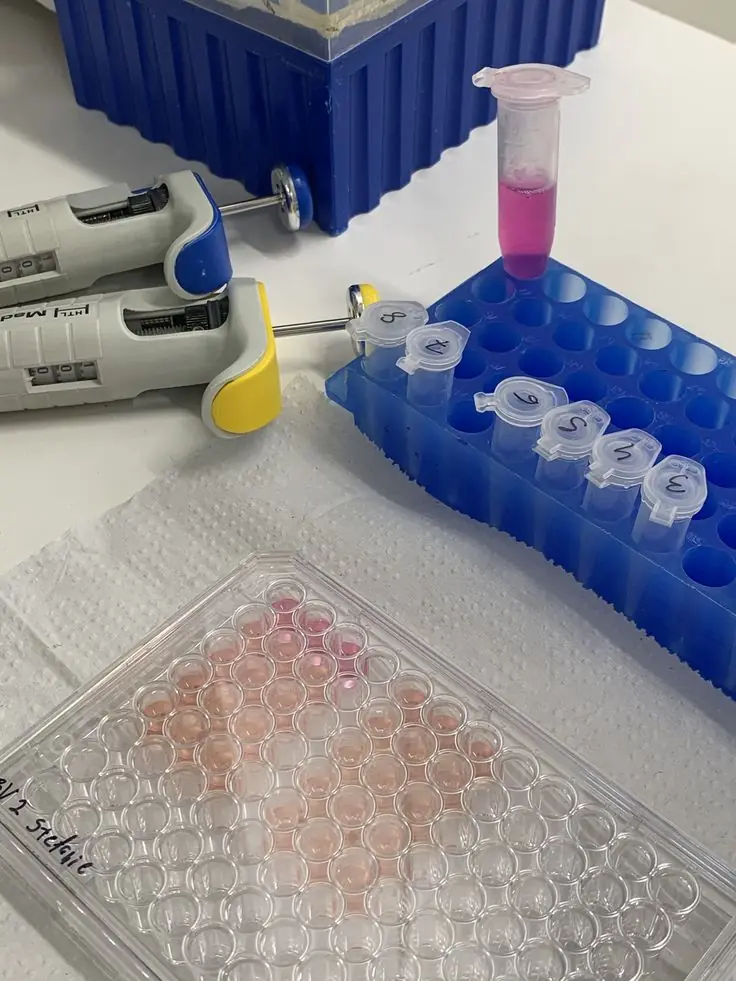
Repeatable handling standards are strongest when they leave little room for interpretation.

Confirmation screens, item summaries, shipping fees, and customer lookup all shape operational confidence.
01
Evidence first. Every note starts with observable product, order, or laboratory behavior.02
Plain systems. No mystique around workflows. The best operations are legible.03
Clean records. Good documentation keeps the storefront, support desk, and lab aligned.